12月19日,Madrigal Pharmaceuticals宣布其治疗NASH的产品Resmetirom(THR-β激动剂)在关键3期临床试验中取得积极顶线结果[1]。肝脏活检数据表明:Resmetirom可显著改善NASH症状和纤维化。组织学疗效是FDA评估NASH新药临床获益的重要参考依据,Resmetirom有望成为FDA批准的第一款NASH治疗药物。Resmetirom的成功也将促进非侵入性诊断疗法在NASH领域中的开发与应用。
 图片来源:Madrigal公司官网
图片来源:Madrigal公司官网
此外,Akero在研的Efruxifermin(Fc-FGF21融合蛋白)临床2期效果优异 [2],近期被FDA授予突破性疗法认定(BTD)[3]。正大天晴与Inventiva合作开发和商业化泛PPAR小分子激动剂Lanifibranor,该药物此前也在美国获得BTD[4]。
一直以来,NASH以其患病群体庞大、尚无上市药物、市场空间大等特点广受各界关注,国内外药企纷纷布局投入研发。然而由于致病机理复杂,新药获批门槛高等限制,纷纷折戟,NASH新药研发一波三折,市场遇冷。今年创新药捷报频发,无疑是为NASH领域的研发者注入了一剂强心针。
疾病的机制研究与药物评估离不开动物模型。此前美国FDA发布的NASH药物开发指导草案中也提到:鼓励研发者使用动物模型进行新药筛选[5]。
维通利华目前可提供NASH现货模型,模拟NASH病理生理学和新陈代学特点,节省研发者前期造模时间和精力,尽快开展研究。
维通利华使用C57BL/6雄性小鼠,从6周龄起连续饲喂特殊饲料(高脂高胆固醇高果糖)至少20-26周,小鼠肝脏呈现NASH典型组织学特点:肝细胞脂肪变性、气球样变和炎症;随着诱导时间延长,还可观察到组织纤维化。
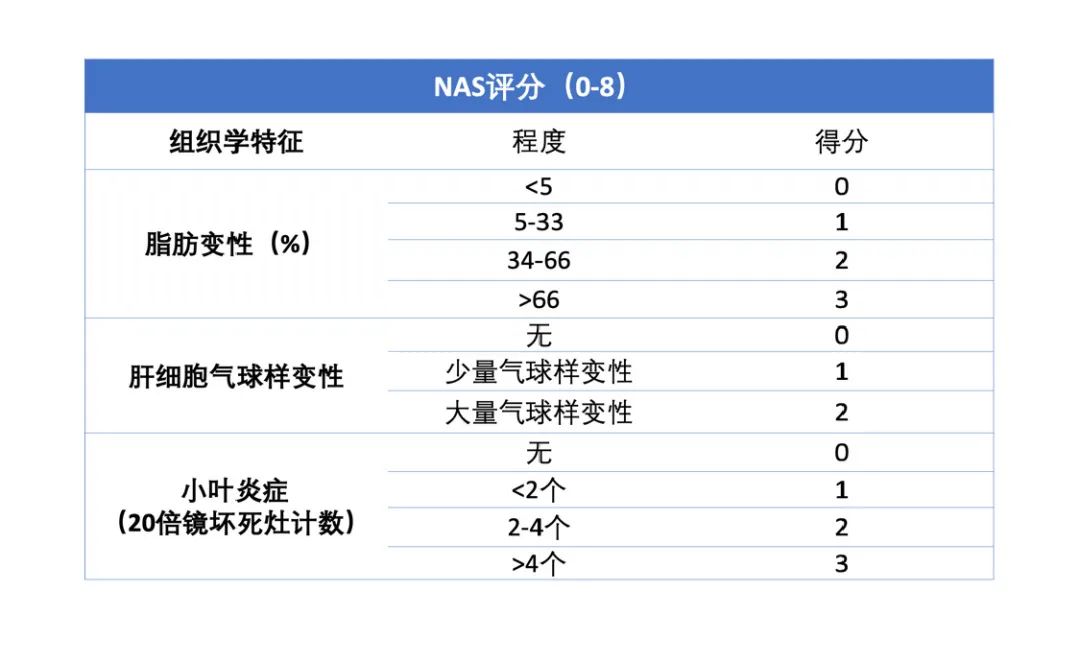
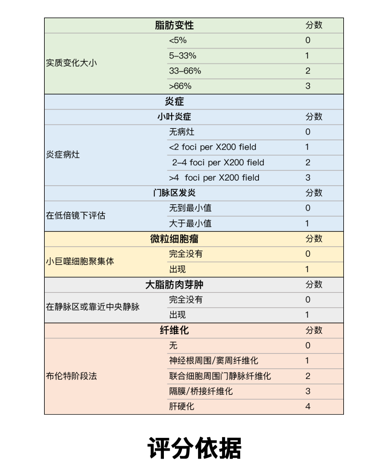
参考NAS score评价模型肝脏组织,数据表明:至少诱导20周起,NAS score≥5,自诱导26周起NAS score稳定在6.5左右,表示造模成功且稳定。
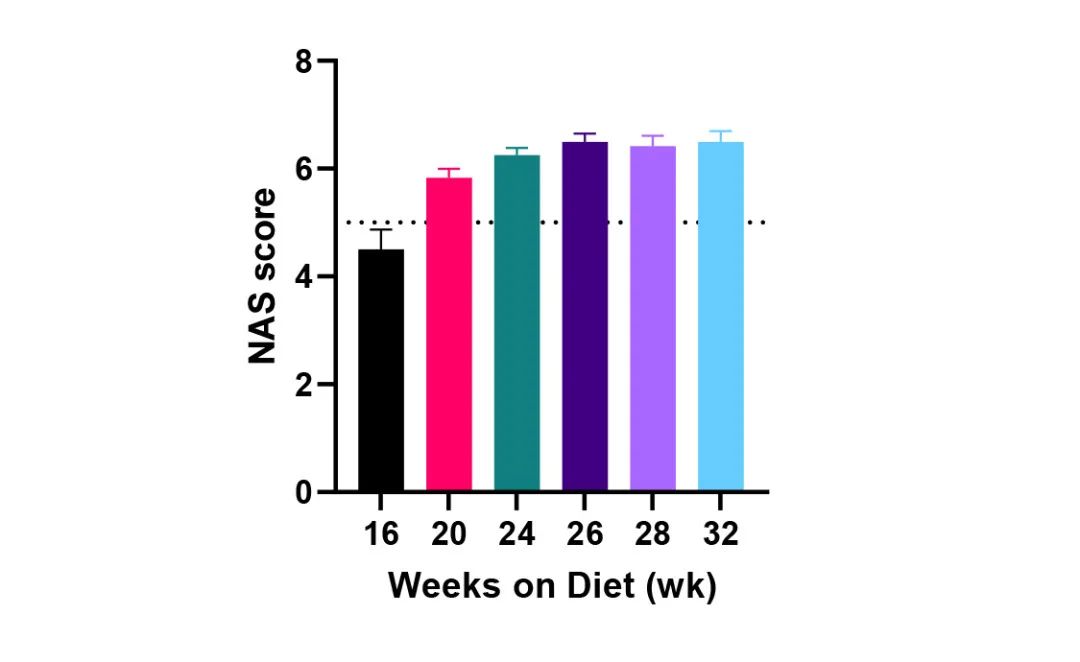
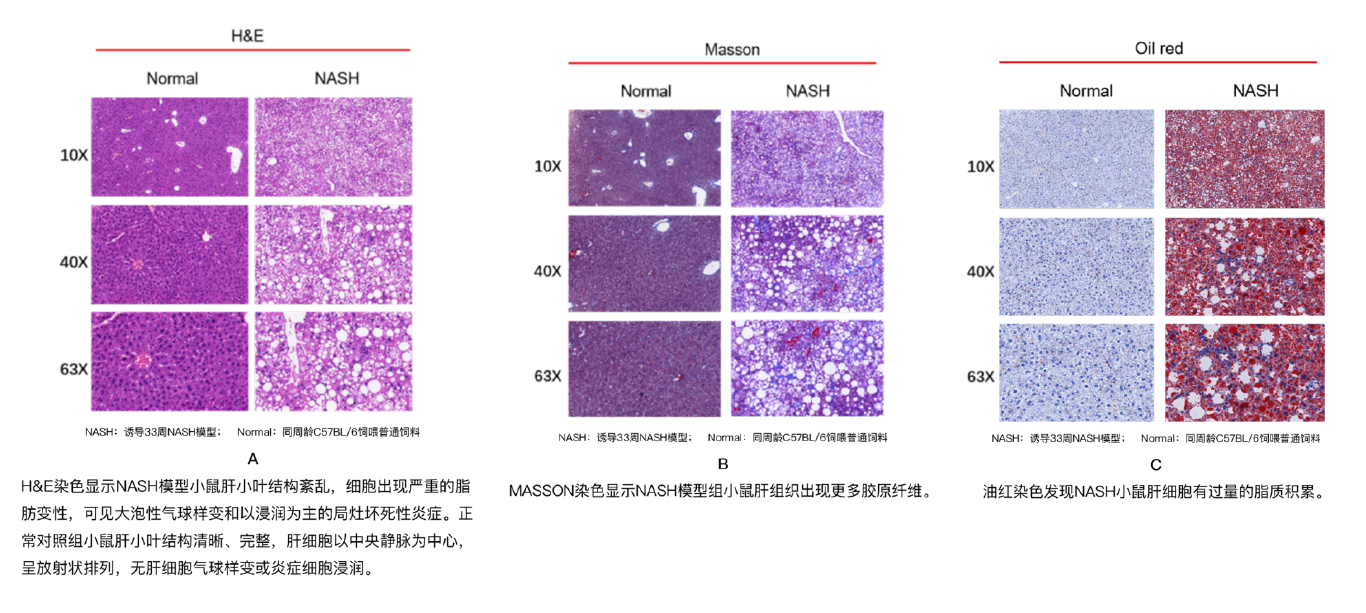

除肝脏组织学高度模拟疾病特征外,NASH模型还具有肥胖、肝脏指数增加、血生化指标异常指向肝肾功能紊乱等临床常见的新陈代谢学特点,多方面模拟NASH疾病进程,助力临床前疾病机制与药效研究。
详情请咨询当地销售
或联系维通利华模型定制服务

参考文献
1. "Madrigal Announces Positive Topline Results from the Pivotal Phase 3 Maestro-Nash Clinical Trial of Resmetirom for the Treatment of Nash and Liver Fibrosis". Madrigal Pharmaceuticals, Inc. 2022-12-19.<https://ir.madrigalpharma.com/news-releases/news-release-details/madrigal-announces-positive-topline-results-pivotal-phase-3>
2."In Akero Therapeutics’ Phase 2b Harmony Study, Both the 50mg and 28mg Efx Doses Achieved Statistical Significance on Primary and Secondary Histology Endpoints after 24 Weeks". Akero Therapeutics,Inc. 2022-09-13.<https://ir.akerotx.com/news-releases/news-release-details/akero-therapeutics-phase-2b-harmony-study-both-50mg-and-28mg-efx>.
3."Efruxifermin Granted FDA Breakthrough Therapy Designation for NASH". Akero Therapeutics,Inc. 2022-12-8.<https://ir.akerotx.com/news-releases/news-release-details/efruxifermin-granted-fda-breakthrough-therapy-designation-nash>
4."Inventiva and Sino Biopharm announce licensing and collaboration agreement to develop and commercialize lanifibranor in Greater China". Inventiva Pharma. 2022-9-21.<https://inventivapharma.com/inventiva-and-sino-biopharm-announce-licensing-and-collaboration-agreement-to-develop-and-commercialize-lanifibranor-in-greater-china/>
5."Noncirrhotic Nonalcoholic Steatohepatitis with Liver Fibrosis: Developing Drugs for Treatment; Draft Guidance for Industry; Availability." Federal Register, 2018. 62582-83. Vol. 83.




